Part 2 of 3
At the beginning of Thanksgiving week of 1989, when these events were taking place at the Reston Primate Quarantine Unit, Thomas Geisbert was a twenty-seven-year-old civilian researcher working at usamriid while he studied for a Ph.D. in microbiology. His specialty is the electron microscope. Geisbert is something of a loner, a tall man with blue eyes, brown hair, and arrestingly large ears. He grew up an only child in western Maryland, where he spent a lot of time camping in the woods alone or with his uncles, who taught him how to hunt and fish. Geisbert’s boss at usamriid was Peter Jahrling. Tom Geisbert goes deer hunting in West Virginia every year around Thanksgiving. He planned to leave on Monday morning of that week, but something prompted him to stop by his lab at usamriid for a last look at the flasks of monkey cells that were incubating the virus from Reston. At nine in the morning, he put on a surgical mask and gloves and entered the BL-3 suite. There he met Joan Rhoderick, the technician who had started the Reston culture. She was looking at a flask under a microscope. The flask contained cells infected with virus from the Reston monkey O53. She said to Geisbert, “There’s something flaky going on in this flask.”
The flask was small—four inches long—and it was made of plastic and had a screw cap. Geisbert looked through the eyepieces of the microscope into the flask. Living cells ordinarily cling to the bottom of a flask in a carpet. This carpet looked eaten by moths. It was full of holes: dead and dying cells had detached from the flask and drifted into the fluid. Later, he described to me what he’d seen. “Cells that have been infected with S.H.F. take on a spiderweb look. These cells didn’t look like that. They were rounded and had a granular, pepperlike look. Some were dead. They were ‘off the plastic,’ as we say. It means they had floated away.”
This didn’t look like simian hemorrhagic fever. He went out and got Peter Jahrling, his boss. He said to Jahrling, “There’s something very strange going on in that flask, but I’m not sure what.”
Jahrling had worked at usamriid long enough to have seen some strange things in flasks. “The cells were blown away. They were crud,” Jahrling recalled later. He thought that a wild strain of bacteria had invaded the cell culture. This is a common and annoying occurrence in cell cultures, and it wipes out the culture. Bacteria give off odors as they multiply, and Peter Jahrling had smelled enough bacterial contaminations so that he knew how to distinguish them by nose. Viruses, on the other hand, kill cells without releasing an odor. Jahrling guessed that the flask had been wiped out by a common soil bacterium named pseudomonas, which, he says, “smells like Welch’s grape juice.” He unscrewed the cap and waved his hand over it, and took a whiff, and said to Geisbert, “Have you ever smelled pseudomonas?” Geisbert accepted the flask from Jahrling and sniffed. He didn’t smell any Welch’s grape juice. There was no odor. Jahrling, who hadn’t smelled anything, either, took back the flask and whiffed it again. Nothing. No smell. But the cells were blown away.
Geisbert poured some milky fluid out of the flask into a test tube and spun it in a microcentrifuge. A small “button” of material collected at the bottom of the test tube—a pill of dead and dying cells. Geisbert removed the button with a wooden stick and soaked it in plastic resin. Then he went hunting in West Virginia. He planned to look at the button in his microscope when he returned, after Thanksgiving. When Ebola virus infects a human being, the incubation period is from seven to fourteen days, while the number of virus particles gradually climbs in the bloodstream. Then comes the headache.
The first known emergence of a filovirus happened in August, 1967, in Marburg, Germany. A shipment of green monkeys from Uganda had arrived in Frankfurt. Green-monkey kidney cells are useful for the production of vaccines, and these monkeys were going to be killed for their kidneys. Most of the monkeys were trucked from Frankfurt to a factory in Marburg that produced serum and vaccines, while a few monkeys from the same shipment stayed in Frankfurt, and a few others went to Belgrade, Yugoslavia. The first person known to be infected with the virus—the index case—was a man known as Klaus F., an animal-care technician at the serum factory in Marburg. He broke with fever and rash on August 8th, and died two weeks later.
So little is known about the Marburg agent that only one book has been published about it, “Marburg Virus Disease,” edited by G. A. Martini and R. Siegert. In it we learn:
The monkey-keeper heinrich p. came back from his holiday on August 13th 1967 and did his job of killing monkeys from August 14th-23rd. The first symptoms appeared on August 21st.
The laboratory assistant renate l. broke a test-tube that was to be sterilized, which had contained infected material, on August 28th, and fell ill on September 4th 1967.
And so on. Thirty-one laboratory workers acquired the disease; seven died. In other words, the case-fatality rate of Marburg virus in hospitalized patients was twenty-two per cent. That was terrifying. Yellow fever, which is considered a lethal virus, kills only five per cent of the infected once they reach a hospital.
Marburg began with a splitting headache, focussed behind the eyes and temples. That was followed by a fever. The characteristic diagnostic sign was a red speckled rash over the body which blistered into a sea of tiny white bubbles. “Most of the patients showed a sullen, slightly aggressive, or negativistic behavior,” Martini wrote. “Two patients [had] a feeling as if they were lying on crumbs.” One became deranged and psychotic. These mental signs were caused by the virus’s having damaged the brain. The patient Hans O.-V. showed no signs of mental change, but he suffered a sudden, acute fall of blood pressure and died. At autopsy, his brain was found to be laced with hemorrhages, and there was a massive, fatal hemorrhage at the center. In Frankfurt, an animal attendant known as B. developed a high fever and eventually began bleeding from his mouth, nose, and gastrointestinal tract. He was given whole-blood transfusions, but then he developed uncontrollable hemorrhages at the sites of the I.V. punctures. He died with blood running from his mouth and his nipples. All the survivors lost their hair. During convalescence, the skin peeled off their faces, hands, feet, and genitals. It was a small, frightening emergence.
Marburg virus looks like rope, or it rolls up into the rings that resemble Cheerios. Virologists had never seen a ring-shaped virus, and couldn’t figure out how to classify it. They thought that it might be a type of rabies. The rabies particle is shaped like a bullet, and if you stretch a bullet it becomes a rod, and the rod can be bent into a doughnut: Marburg. They started calling Marburg “stretched rabies.” But it is not related to rabies.
The question was: What is the virus’s natural history? In what animal or insect does Marburg hide? Marburg evidently does not circulate in monkeys. Monkeys die quickly of the disease, and if they were the reservoir, Marburg wouldn’t wipe them out. The monkey’s immune system would have learned to attack the virus, and the virus itself would have become better adapted to living in monkeys without killing them, since it is in the virus’s best interest to let the host survive. The Marburg monkeys had been collected in Uganda by native trappers—apparently in forested habitat to the west of Mt. Elgon, an extinct volcano that straddles the border between Uganda and Kenya. Teams of epidemiologists combed Uganda, and especially the western slopes of Mt. Elgon, looking for some animal or insect that harbored Marburg virus; they found nothing.
In 1980, a French engineer who was employed by the Nzoia Sugar Company at a factory in Kenya within sight of Mt. Elgon developed Marburg and died. He was an amateur naturalist who spent time camping and hiking around Mt. Elgon, and he had recently visited a cavern on the Kenyan side of the mountain which was known as Kitum Cave. It wasn’t clear where the Frenchman had picked up the virus, whether at the sugar factory or outdoors. Then, in the late summer of 1987, a Danish boy whose name will be given here as Peter Cardinal visited the Kenyan side of Mt. Elgon with his parents—the Cardinals were tourists—and the boy broke with Marburg and died.
Epidemiologists at usamriid became interested in the cases, and they traced the movements of the French engineer and the Danish boy in the days before their illnesses and deaths. The result was weird. The paths of the French engineer and the Danish boy had crossed only once—in Kitum Cave. Peter Cardinal had gone inside Kitum Cave. As for the Ugandan trappers who had collected the original Marburg monkeys, they might have poached them from the Kenyan side of Mt. Elgon. Those monkeys might have lived near Kitum Cave, and might even have occasionally visited the cave.
Mt. Elgon is a huge, eroded volcanic massif, fifty miles across—one of the largest volcanoes in East Africa. Kitum Cave is one of a number of caverns that penetrate Mt. Elgon at an altitude of around eight thousand feet and open their mouths in a deep forest of podo trees, African junipers, African olives, and camphors. Kitum Cave descends into tight passages and underground pools that extend an unknown distance back into Mt. Elgon. The volcanic rock within Kitum Cave is permeated with mineral salts. Elephants go inside the cave to root out chunks of salty rock with their tusks and chew on them. Water buffalo also visit the cave to lick the rocks, and they may be followed into the cave by leopards. Fruit bats and insect-eating bats roost in the cave, filling the air with a sour smell. The animals drop their dung in the cave—an enclosed airspace—and they attract biting flies and carry ticks and mites. The volcanic rock contains petrified logs, the remains of trees that were enveloped in lava, and the logs are filled with sharp crystals. Peter Cardinal may have handled crystals inside the cave and scratched his hands. Possibly the crystals were tainted with animal urine or the remains of an insect. The Army keeps some of Peter Cardinal’s tissues frozen in cryovials, and the Cardinal strain is viciously hot. It kills guinea pigs like flies. In February, 1988, a few months after Peter Cardinal died, the Army sent a team of epidemiologists to Kitum Cave.
The team wore Racal suits inside the cave. A Racal is a lightweight pressurized suit with a filtered air supply, used for hot operations in the field. There is no vaccine for Marburg, and the Army people had come to believe that the virus could be spread through the air. Near and inside the cave they set out, in cages, guinea pigs and primates—baboons, green monkeys, and Sykes’ monkeys—and they surrounded the cages with electrified wire to discourage predators. The guinea pigs and monkeys were sentinel animals, like canaries in a coal mine: they were placed there in the theory or the hope that some of them would develop Marburg. With the help of Kenyan naturalists, the Army team trapped as many different kinds of wild mammals as they could find, including rodents, rock hyraxes, and bats, and drew blood from them. They collected insects. Some local people, the il-Kony, had lived in some of the caves. A Kenyan doctor from the Kenya Medical Research Institute, in Nairobi, drew blood from these people and took their medical histories. At the far end of Kitum Cave, where it disappears in pools of water, the Army team found a population of sand flies. They mashed some flies and tested them for Marburg.
The expedition was a dry hole. The sentinel animals remained healthy, and the blood and tissue samples from the mammals, insects, arthropods, and local people showed no obvious signs of Marburg. To this day, the natural reservoir of Marburg is unknown. Marburg lives somewhere in the shadow of Mt. Elgon.
On July 6, 1976, five hundred miles northwest of Mt. Elgon, in the township of Nzara, Sudan, in densely wooded country at the edge of the African rain forest, a man referred to as YuG died of a hemorrhagic fever. He was a storekeeper in a cotton factory, and he was the index case of a new strain of filovirus. The clinical features of the disease were indistinguishable from those of Marburg—masklike facial expression, rash, bleeding, terminal shock. Two of YuG’s co-workers also came down with the disease and died. No one knows how the virus got into the cotton factory. One of the dead men, a man known as PG, had a wide circle of friends and contacts, and he also had several mistresses. Most of the subsequent fatal cases of what later came to be known as the Sudan subtype of Ebola hemorrhagic fever can be traced back through chains of infection to PG, through as many as six generations of infection. The strain burned through the town of Nzara, and then reached eastward to the town of Maridi, where there was a large hospital, and it hit the hospital like a bomb. It killed nurses and aides, and it savaged patients and then radiated outward from the hospital through patients’ families. (A characteristic of a lethal, highly transmissible, and incurable virus is that it kills medical people first. Frequently, as in this case, the medical-care system actually intensifies the outbreak, like a lens that focusses sunlight in a heap of tinder.) The Sudan virus was more than twice as lethal as Marburg—its case-fatality rate was fifty per cent, the same as that of bubonic plague before antibiotics. And the death rate kept climbing, until by the third month of the Sudan outbreak mortality among the infected had hit seventy per cent, as if perhaps the virus were mutating, getting hotter as it passed from generation to generation in humans. Then, for reasons that aren’t clear, the outbreak subsided. The surviving staff of the Maridi hospital had panicked and run away, and that may have helped break the chain of infection. Or possibly the human hosts died too quickly to be efficient transmitters of the virus. Whatever the reason, the organism vanished.
In early September, 1976, two months after the beginning of the Sudan break, a similar yet more lethal strain emerged five hundred miles to the west, in the Bumba Zone of Zaire, an area of humid rain forest drained by the Ebola River. The Ebola River strain seemed to come out of nowhere, and popped up in the Yambuku Mission Hospital, an upcountry clinic run by Belgian nuns. The nuns and staff at Yambuku were using five needles a day to give injections of antibiotics and vitamins to hundreds of people in the hospital’s outpatient and maternity clinics. The staff sometimes rinsed the needles in a pan of warm water between injections. The virus entered the cycle of dirty needles, and erupted in fifty-five villages around the hospital. It first killed people who had received injections, and then killed family members—particularly women, who in Africa prepare the dead for burial.
The virus also wiped out the Yambuku hospital’s medical staff. (Medical people go first.) By the end of September, two-thirds of the staff were dead or dying, and the hospital closed down. A critically ill Belgian nun who was a nurse at the hospital, Sister M.E., was flown to Kinshasa, the capital of Zaire, with another nun, Sister E.R., who nursed her. Sister M.E. was admitted to the Ngaliema Hospital, and she died there shortly afterward. Sister E.R. then became ill and died. Then a Zairian nurse at Ngaliema Hospital, identified as M.N., developed fever and bleeding. She had cared for Sister M.E.; she herself would soon die. While M.N. was incubating the virus, she had had face-to-face contact with several dozen people in the city of Kinshasa. The virus seemed about to start an explosive chain of lethal transmission in Kinshasa, a poor, crowded city with a population of two million, where the virus might go off like a bonfire. This epidemiological possibility triggered a panic in European capitals. Kinshasa has direct air links to Europe, and European governments contemplated blocking flights from Kinshasa. The World Health Organization feared that the nurse M.N. might be the vector for a worldwide pandemic. The Zairian government ordered its army to seal off the Bumba Zone with roadblocks, and all radio contact with the province was lost. Bumba had dropped off the earth, into the silent heart of darkness.
Out of Bumba came some tubes of blood, and from Sudan came some vials of serum. A few of the samples ended up in Atlanta, Georgia, at the Centers for Disease Control, where a team headed by Karl M. Johnson isolated the Ebola River virus for the first time. Key members of the team were Frederick A. Murphy, who is an expert in the electron microscope, and Patricia A. Webb, a virologist. (She was married to Karl Johnson at the time.) The team started to grow the virus in cultures of monkey cells, and Murphy began looking at the cells in his microscope. On October 13th, Webb telephoned her husband, Johnson, and said to him, “Karl, you’d better come quick to the lab. Fred has harvested some cells, and they’ve got worms.” The virus looked like Marburg, but Johnson found that it didn’t react to Marburg antibodies. Therefore it was a new virus. Karl Johnson and his team had performed what is known as the first isolation and characterization of the agent—they had got it to replicate, and they had proved it was something new. (Teams at the Microbiological Research Establishment in Porton Down, England, and at the Institute for Tropical Medicine in Antwerp, Belgium, had isolated the virus, too, but they didn’t know what it was.) Johnson’s team had earned the right to name the organism. They named it Ebola.
I learned that Johnson could be reached at a fax number in Big Sky, Montana, so I sent him a fax, in which I said that Ebola virus fascinated me. My fax machine emitted this reply:
Mr. Preston:
Unless you include the feeling generated by gazing into the eyes of a waving confrontational cobra, “fascination” is not what I feel about Ebola. How about shit scared?
The richest trout river in America may be the Bighorn, a green, muscular river in Montana that flows out of the Bighorn Mountains into grassland, and is lined with cottonwoods. One recent day in October, the brown trout were spawning in the Bighorn, and the cottonwoods had turned yellow and rattled in a south wind. Standing waist-deep in a mutableslick of the river, wearing sunglasses, with a cigarette hanging from the corner of his mouth and a fly rod in his hand, Karl Johnson ripped his line off the water and laid a cast upstream. Johnson is a great figure in the history of virology; he trained an entire generation of field virologists at a tropical laboratory called MARU, which he ran in Panama. “I’m so glad nature is not benign,” he said. He studied the water, took a step downstream, and whipped another cast. “But on a day like today, we can pretend nature is benign—all monsters and beasts have their benign moments.” Johnson was a member of a World Health Organization team that went to Kinshasa to try to contain the Ebola virus. “When we got to Kinshasa, the place was an absolute madhouse. There was no news coming out of Bumba province, no radiocontact. We knew it was bad in there, and we knew we were dealing with something new. We didn’t know if the virus could be spread by droplets in the air, somewhat like influenza. If Ebola had easily spread through the air, the world would be a very different place today.”
“How so?”
“There would be a lot fewer of us. It would have been exceedingly difficult to contain that virus if it had had any major respiratory component.”
“Were you afraid you wouldn’t come out alive?”
“Yeah. But I’d been there before. In 1963, I led the investigation of the Machupo outbreak, named for a river that runs by a little town in the plains of eastern Bolivia. Same kind of thing. People bleeding and dying.”
Karl Johnson performed the first isolation of the Machupo virus, a deadly emerger that belongs to a family known as the arenaviruses, because the virus particles are speckled with dots that look like sand. (Arena is Latin for “sand.”) Johnson came down with Machupo in Bolivia—he went into borderline shock in a hospital in the Canal Zone, after he’d been flown out of Bolivia, and he nearly died. Johnson also collaborated on the first isolation of the Hantaan virus, a lethal east-Asian organism (classified as a BL-3 agent), which happens to be another important emerger. A Hantaan relative now infects the rats of Baltimore and Philadelphia; no obvious human epidemic has yet occurred in the United States. Johnson has therefore been credited with work that led to the discovery and classification of three major groups of emerging human hemorrhagic-fever viruses—the filoviruses, the arenaviruses, and the hantaviruses (named after Hantaan).
“I’ve seen young physicians run from these hemorrhagic viruses, literally,” he said. “In the Zaire thing, we had a young doctor from the C.D.C. who just couldn’t get on the plane with me to Kinshasa. He admitted he was too afraid. We sent him home. I did figure that if Ebola was the Andromeda strain—incredibly lethal and spread by droplet infection—then there wasn’t going to be any safe place in the world anyway. It was better to be working at the epicenter than to get the infection at the London opera.”
The W.H.O. team in Zaire wore fabric helmets with full-face respirators, and disposable gowns, gloves, and overshoes. They set up two containment pavilions at Ngaliema Hospital. Into one pavilion they shut thirty-seven people who had had face-to-face contact with M.N., the Zairian nurse who was then dying, and into the other pavilion they shut all medical staff who had had contact with the nuns who had already died. Doctors and nurses entered the containment areas through a double-doored antechamber, a gray zone. They wrapped the cadavers of the nuns and the nurse (when she died) in sheets soaked in a phenolic disinfectant, then double-bagged these mummies in plastic, put them in coffins that had screw-down lids, and issued instructions to the families of the deceased to bury the coffins immediately, with no wake. The rooms where the nuns had suffered their agonals were not pleasant to behold. The floors, furniture, and walls were stained with blood. The aspect of those rooms may have raised in some minds one or two questions about the nature of the Supreme Being; or, for persons not inclined to theology, the blood on the walls may have served as a reminder of the nature of Nature. The team washed everything with bleach and smoked the victims’ rooms with formaldehyde vapor. No one in the containment pavilions or in the city fell ill with the virus. Somewhat to the team’s surprise, and to its great relief, the Ebola agent seemed not to be contagious in face-to-face contacts.
“We got an advance party into the bush with a couple of Land Rovers,” Johnson said. “They wore respirators and paper gowns and rubber gloves. It turned out that the epidemic was already in decline when the teams got there. The village elders had had the wisdom to institute procedures for dealing with smallpox, which has been a problem for centuries in Africa. An infected person was put in a hut by himself, and food and water were pushed through the doorway. If the person was able to care for himself, he’d eventually come out of the hut. Otherwise, they’d burn the hut down. It really worked with Ebola. But think what that does to a traditional culture. In order to stop an epidemic that way, you have to suspend all the normal cultural relations that surround death. You have to put a parent or a child into that hut and burn it down afterward. The African technique would work in the United States, but I don’t think we’d do it.”
During Thanksgiving week of 1989, Nancy Jaax’s father was dying of cancer in Wichita, Kansas, and she and Jerry drove home. Nancy had grown up on a farm in Wichita. Her father had owned a small chain of hamburger restaurants called Dunn’s Grills. They lived on a farm outside town, where they grew truck crops, such as tomatoes, cantaloupes, peppers, watermelons, and corn, for the restaurants. Nancy would get up at five in the morning to work in the fields with her father. Later, in high school, she moved in with her grandmother in Wichita, and in the evenings she would help run another restaurant owned by her father called the Plantation (her father had sold Dunn’s Grills). Thanksgiving of 1989 was the most painful family reunion of her life. She said her farewell to her father. She didn’t know whether she would see him again.
Tom Geisbert shot a buck in West Virginia, and returned home to spend Thanksgiving with his family. Dan Dalgard spent an uneasy Thanksgiving with his wife. He had not stopped the apparent course of simian hemorrhagic fever in his monkeys by sacrificing the monkeys in Room F. Dead monkeys appeared in Room H, two doors down the hall from Room F. After the holiday weekend, Dalgard performed necropsies on four monkeys, taking slices of spleen, liver, and kidney. He wrote in his diary, “Gut feeling after looking at the animals and tissues is that we are not seeing lesions compatible with S.H.F.” He had no idea what was killing his monkeys.
At seven-thirty on Monday morning, November 27th, Tom Geisbert reported to work at his laboratory at usamriid. He wanted to get an early start with his electron microscope, looking at the button of dead cells he had harvested the previous Monday. Recently, I met with Geisbert in his office. The walls were plastered with photographs of the Ebola virus. Some of the viruses were ten inches long and resembled ballpark frankfurters. I asked him how he takes a photograph of a virus. He unlocked a filing cabinet and removed from it a metal object the size of a pocket pencil sharpener. “This is a diamond knife,” he said. “These things cost about four grand apiece. See the diamond?” Hesitantly, he slid his treasure across his desk toward me, and I picked it up. A prism gleamed. “Please don’t touch the edge,” he said. “You’ll completely trash it. You’ll dull it, and your finger oils will stick to the edge. Four thousand dollars.”
He showed me a button of cells. It was a dot the size of a toast crumb, embedded in a wedge of clear plastic. The cells—from a monkey’s liver—were almost rotten with Ebola virus, but he’d sterilized the button with chemicals. He took the button into another room, where he mounted the button and the diamond knife in a machine and threw a switch. The machine worked like a deli slicer. It drew the diamond knife across the button, peeling off a slice, just like a slice of luncheon meat. The slice was this size:
•
It contained as many as ten thousand cells. Geisbert picked up the slice with a tiny copper mesh, and carried the sample into a darkened room containing a metal tower taller than a person. That was his microscope. He put the sample in a chamber in the microscope, and pushed a button. A complicated image appeared on a viewing screen, showing a tiny corner of one cell—a cellscape of oxbow rivers and lakes that reminded me of an aerial view of jungle.
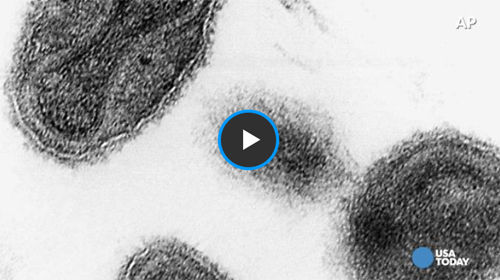
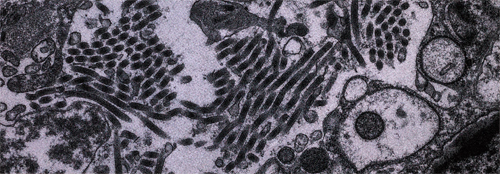
“I don’t see any Ebola here,” Geisbert declared, turning a knob, while the cellscape drifted across the field of view. We huddled over the viewing screen, and lakes and paths and specks went by almost without end, until I felt as if we were inside a starship, making a low-orbit pass over a huge, unexplored planet near Tau Ceti. “Sometimes the viruses are everywhere, or sometimes I have to look for six hours before I find a particle,” Geisbert said. He was immutably patient, his eyes scanning the terrain. He could pick out patterns of sickness in a cell, subtle anomalies which, like footprints, would lead him to the horrible brood. In the case of Ebola, it is a brood. When Ebola replicates, the virus grows in blocks inside a cell, which are like nests. These are the inclusion bodies, or bricks. The bricks migrate toward the surface of the cell. As a brick reaches the cell wall, it disintegrates into hundreds of individual viruses, and the broodlings bud through the cell membrane and float away in the universe of the host. No one knows how the Ebola bricks are propelled toward the surface of the cell.
“That was quite a day,” Geisbert said, sitting at the microscope in the darkened room. His face glowed in the light of the screen. “It’s in the morning, around ten o’clock. The sample is cell culture from Monkey O53. I put the sample in the scope. I switch it on. I’ve looked at it for maybe fifteen seconds, and then—‘Oh, shit.’ The tissue was a mess, and it was wall-to-wall with filovirus.” Some areas were so thick with virus that they looked like buckets of rope. “I almost lost it,” he said. “The only filovirus I’d ever seen in the microscope was Marburg. I had worked with the Cardinal strain of Marburg—the strain from the Danish boy who got Marburg at Kitum Cave in Mt. Elgon—and I knew what that looked like. So I thought, Marburg. I knew that Pete Jahrling and I had sniffed those flasks. I thought, Oh, man, Pete and I have been handling this stuff in BL-3 conditions, and this is a BL-4 agent.”
He developed a few photographs of the virus particles and hurried into the office of Peter Jahrling, his boss. Jahrling reacted calmly. It seemed to be a filovirus—Jahrling could see wormlike shapes. Jahrling and Geisbert could have breathed it into their lungs. They began counting days back to the time of their exposure. Seven days had passed since they inhaled from the flask. Well, they didn’t have headaches yet.
Jahrling went to get his boss, Colonel Clarence James Peters—he goes by the name C.J.—who was then the chief of the disease-assessment division at usamriid. Colonel Peters came into Jahrling’s office and looked at Geisbert’s photographs. Peters feared that any public announcement of a Marburg-virus outbreak might cause a panic in Reston, once people had learned the history of Marburg. He wanted to get a definite positive identification of the strain before the Army made any announcement.
Tom Geisbert stayed up most of that night. He went into the BL-3 laboratory and found a plastic jug that contained sterilized pieces of liver from Monkey O53. He fished some liver out of the jug, clipped bits off it, and fixed the bits in plastic, preparatory to slicing them for viewing in his electron microscope. He left the plastic to cure and went home for a couple of hours to try to sleep. He returned to Fort Detrick while it was still dark, at five in the morning, and before the sun rose he had developed photographs of filovirus particles budding directly out of cells in the monkey’s liver. It was a definite confirmation that the Reston monkeys were infected with a filovirus. But what strain was it? Everyone assumed that it was Marburg, which kills about one in four people it infects. All that day, in his laboratory, Peter Jahrling used a fluorescence test to try to nail down the strain. At five o’clock in the evening, he put some samples under an ultraviolet light and, to his shock, found that the stuff that glowed wasn’t Marburg: it was Ebola, the slate-wiper, which kills almost nine out of ten people.
The news that Ebola virus had broken out near Washington, D.C., was not received casually at Fort Detrick. Shortly after five o’clock, minutes after Jahrling typed the strain, Colonel Peters notified the chain of command. First, Peters and Jahrling went to Colonel David Huxsoll, the head of usamriid. Picking up Huxsoll and then Nancy Jaax, the group then went to Major General Philip Russell, the commander of the Army Medical Research & Development Command at Fort Detrick. General Russell was himself a virologist, and when he saw Geisbert’s glossy photographs he knew what he was looking at. The meeting became tumultuous. With people talking loudly in the background, General Russell picked up the telephone and called the Centers for Disease Control, and got Frederick Murphy on the line. Murphy is an expert on the Ebola virus—he had performed the first isolation of the virus with Karl Johnson, during the 1976 Zaire outbreak—and now, perhaps understandably, Murphy was skeptical when General Russell told him that the Army had isolated Ebola near Washington. Murphy is reported to have said to General Russell, “You can’t fool me. You have crud in your scope.” Still, Murphy took it seriously. He said that a team from the C.D.C. would fly to usamriid early the next morning to review the data. He advised Russell to notify Hazleton Research Products, so that the company’s employees could be protected, and also to notify the Virginia State Department of Health.
Russell and Huxsoll put C.J. Peters in charge of any Army units that would be needed to deal with the Ebola outbreak. Next, Peters set up a conference call with Dan Dalgard, at his home. He told Dalgard that his monkeys had Ebola virus, probably in a mixed infection with simian hemorrhagic fever. Dalgard had heard of Marburg but never of Ebola.
The next morning—Wednesday, November 29th—seven dead monkeys turned up in Room H at the Reston Primate Quarantine Unit. It seemed that Room H had now become the hot spot.
Then Dalgard got another disturbing piece of news. An animal caretaker at the Reston monkey unit, who will here be called Jarvis Purdy, had suffered a heart attack and had been taken to Loudoun Hospital, near Reston. Dalgard wondered if Purdy’s heart attack had been triggered by an Ebola infection. Had Purdy thrown an Ebola clot? Dalgard called the hospital and, without mentioning the word “Ebola,” left instructions for Purdy’s doctor that if he saw any unusual signs in Purdy he should immediately notify Colonel C. J. Peters, of the United States Army. Dalgard also issued an order to the monkey caretakers at the Reston unit. As he recorded in his journal,
All operations other than feeding, observation and cleaning were to be suspended. Anyone entering the rooms was to have full protection—Tyvek suit, respirator, and gloves. Dead animals were to be double-bagged and placed in a refrigerator.
That morning, Colonel Peters and Lieutenant Colonel Nancy Jaax drove down to Hazleton Washington’s headquarters, in Vienna, where Dalgard has his office and the company has a laboratory. Peters, in command of the Army groups that would respond to the Reston emergence in whatever way might be needed, sensed that the Army might have to act decisively to deal with the virus. As he drove to Vienna, he turned over in his mind the question of whether the Army would have to sterilize the Reston Primate Quarantine Unit, using military biohazard teams. There is a slang term in the Army for this type of action: the term is “nuke.” In the world of biocontainment, nuke has nothing to do with nuclear weapons. It has to do with neutralizing hot organisms: to nuke a place means to sterilize it. You go into the place in space suits and you isolate any infected hosts. If the hosts are animals, you kill them, bag them, and incinerate them. If the hosts are human, you put them in bubble stretchers and take them to the biocontainment hospital at usamriid—the Slammer. Then you sterilize the hot zone with biocides and formaldehyde gas.